Monitoring Software
The Rotronic Monitoring System
The Rotronic Monitoring System (RMS) is a GAMP©5 category 4 software combined with category 1 hardware, helping users monitor their GxP compliant applications, looking into the critical quality attributes and monitoring critical process parameters, helping focus on patient safety, product quality and data integrity and compliant to EudraLex Annex 11 and FDA 21 CFR Part 11.
With RMS, monitoring of any parameter is simple with the core RMS hardware, but 3rd party integration is also possible via analogue and MODBUS TCP signals. Integration and extraction of data is also possible with RMS’s application programming interface (API).

Designed with pharmaceutical companies for pharmaceutical companies, the user-friendly interface makes the monitoring so easy, that the user only has to focus on their main tasks.

Your main benefits:
- Compliance, audit and risk management
- Time saving with automatic data collection and report generation
- 24/7 real-time centralised monitoring of all operations
- All critical measurements in one system
- Real time alarming for critical measuring points

Fast & Easy with the Rotronic SaaS Cloud solution:
Forget the hassle of having to maintain your IT infrastructure and focus only on what is crucial to your application. You can setup multiple measurement points in various locations and access the data from any device with a browser and internet connection. Setup alarms and notifications to be sure that you are always informed of what is happening!

Remain compliant to GxP regulations with the Rotronic Exclusive SaaS Cloud solution:
Hosted on a high security and redundant datacentre with your own virtual server, you can run the Rotronic category 4 software in the cloud throughout each and every GxP compliant facility. Equip your worldwide facilities with the RMS hardware and access all of the data, including the audit trail, alarming and data analysis functions from a completely validated system.

Take advantage of your own IT infrastructure with the RMS on-premise solution:
Run the RMS software with an SQL database on your own IT infrastructure and have complete control over your entire system and comply to your own internal regulations, access the interface via most browsers.
The main software features:
- Alarm overview - see directly how many alarms are active and where it is coming from
- Receive alerts via interactive phone, texts and E-Mails
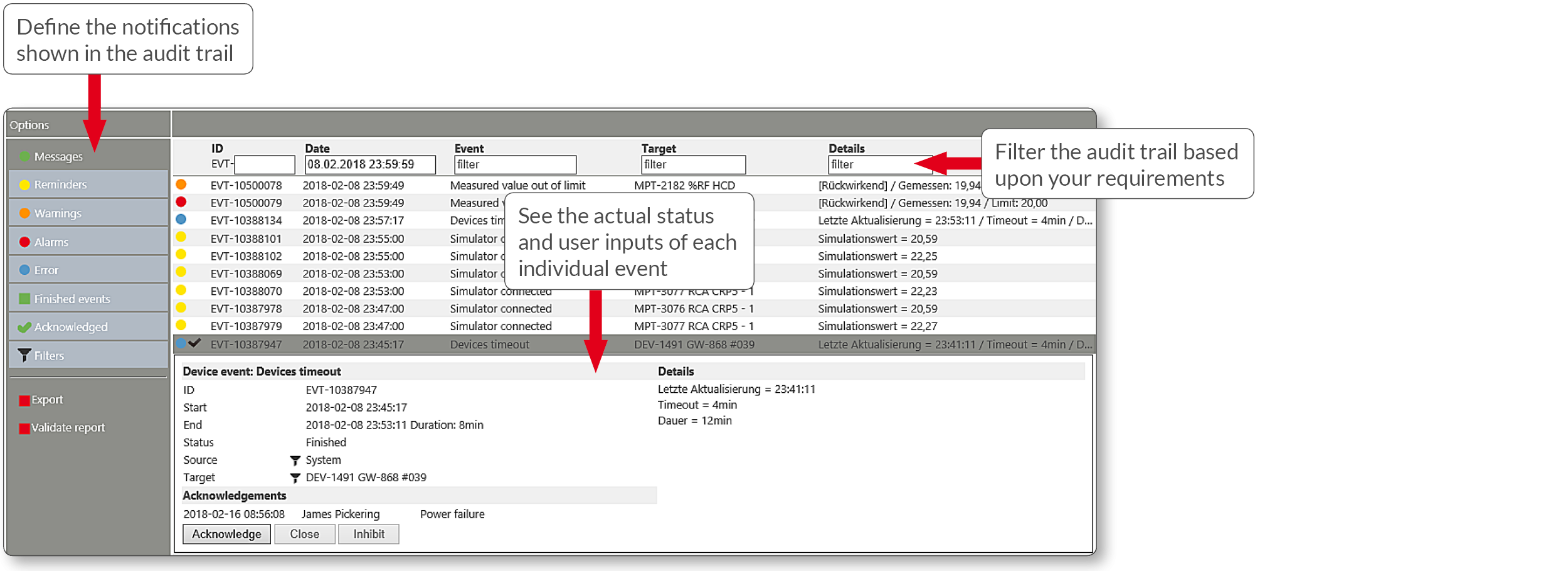
- Audit trail to see exactly what happened and when
- Monitor multiple locations - one system for all your monitoring locations
- Input floor plans - get a real overview of the setup
- Scalable - prefect for single points in small installations to thousands of points in worldwide installations
- Automatic report generation
- Integration of Rotronic and any third party hardware - analogue and digital
- Create custom dashboards per user
- FDA 21 CFR Part 11 & EU Annex 11 complaint
- GAMP©5 conform for GxP applications
- Automatic validation scripts - for fast and effective OQ validation
- Continuously developing based around customer feedback
Additional features:
- Add an IP camera and add a snap shot to your alarm report
- Calibrate all of your measuring points, possibility to adjust all of the Rotronic devices
- Archive all measuring points that are no longer needed, keeping access to the data for as long as necessary
- Save all your documents in the system: user manuals, data sheets, calibration certificates, SOP’s...
Register now for a free demo account!
System overview:

Table View:

Layout View:

User defined dashboard overview:

Audit trail view: