Monitoring in rooms of health care (hospitals, clinics…)
The Rotronic Monitoring System can help with 3 critical applications that require environmental monitoring within the healthcare industry:
1. Statutory/Regulations
2. Clinical
3. Personal comfort
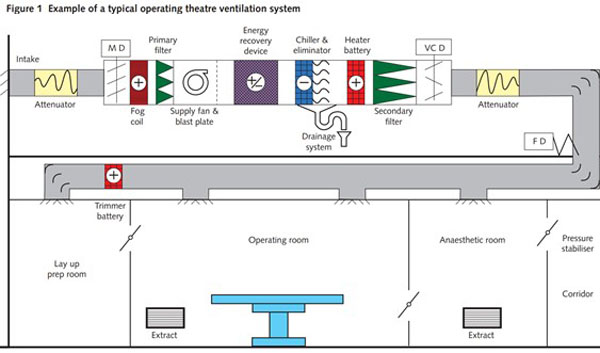
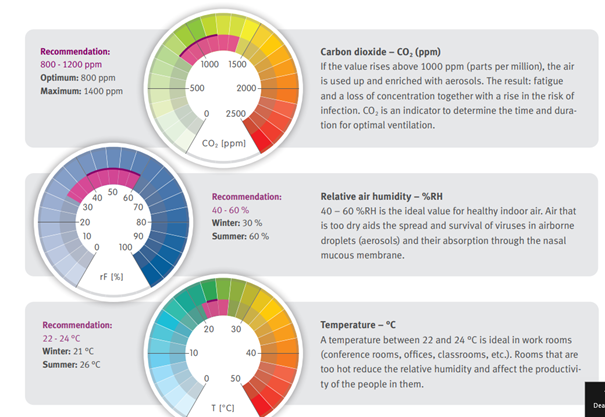
In all healthcare environments (hospitals, clinics…) a critical part of the building management system (BMS) is the heating, ventilation and air conditioning (HVAC). The HVAC manages the air supply within a building to ensure specific environmental conditions for specific areas based upon the application carried out within the area:
- To remove, contain or dilute specific contaminants and fumes,
- To ensure the isolation of one space from another,
- To preserve a desired air-flow path from a clean to a less clean area,
- To provide control of the cleanliness of a space
- To provide close control of temperature,
- To provide close control of humidity…
As air can transport and spread airborne infectious agents it is crucial to ensure that the HVAC is working correctly.
RECOMMENDED PRODUCTS
-
RMS-LOG-T30-915
The RMS-LOG-T30 is a data logger with two integrated analog-to-digital converters, to which two PT100 sensors can be connected for high-precision temperature measurement.
Learn More -
RMS-LOG-T30-868
The RMS-LOG-T30 is a data logger with two integrated analog-to-digital converters, to which two PT100 sensors can be connected for high-precision temperature measurement.
Learn More -
RMS-LOG-T30-L - Data Logger
The RMS-LOG-T30 is a data logger with two integrated analog-to-digital converters, to which two PT100 sensors can be connected for high-precision temperature measurement.
Learn More -
RMS T30-0006
The T30 temperature sensors are resistance thermometers (Pt100 = positive measurement resistance), meaning that the resistance increases with increasing temperature. The sensors are compatible with the RMSLOG- T30-L, RMS-LOG-T30-868 and RMS-LOG-T30-915 data loggers.
Learn More -
RMS-LOG-915 - Data Logger - Wireless Interface - 915 MHz
The flexible dataprotection component of the Rotronic Monitoring System RMS Learn More -
RMS-LOG-868 - Data Logger - Wireless Interface - 868 MHz
The flexible dataprotection component of the Rotronic Monitoring System RMS Learn More -
RMS-LOG-L - Data Logger - LAN Interface
The flexible dataprotection component of the Rotronic Monitoring System RMS Learn More -
PCD-S - RMS Differential Pressure Probe
The compact differential pressure probe with flow or diaphragm sensor technology Learn More -
CCD-S - RMS CO2 Probe
The Compact CO2 Probe
Learn More -
RMS-HCD-S - Humidity and Temperature Probe
The HCD-S is a completely digital probe designed for the Rotronic Monitoring System RMS. It measures humidity and temperature and calculates the dew/frost point.
Learn More